Dry mouth associated with Sjogren’s syndrome
Adult: 30 mg tid.
|
Indications and Dosage
Oral
Dry mouth associated with Sjogren’s syndrome Adult: 30 mg tid.
|
|
Contraindications
Uncontrolled asthma, acute iritis, narrow-angle glaucoma.
|
|
Special Precautions
Patients with cardiovascular disease (e.g. MI, angina pectoris), controlled asthma, chronic bronchitis, COPD, history of nephrolithiasis or cholelithiasis. Pregnancy and lactation. CYP2D6 poor metabolisers.
|
|
Adverse Reactions
Significant: Parasympathomimetic effects (e.g. AV block, bradycardia, cardiac arrhythmia, hypotension, lacrimation, respiratory distress, tachycardia, tremor, vomiting).
Cardiac disorders: Palpitation, chest pain. Gastrointestinal disorders: Nausea, abdominal pain, diarrhoea, salivary gland pain, excessive salivation. General disorders and administration site conditions: Hyperhidrosis, back pain, fatigue, fever, malaise. Metabolism and nutrition disorders: Loss of appetite. Musculoskeletal and connective tissue disorders: Arthralgia, skeletal pain. Nervous system disorders: Headache. Psychiatric disorders: Insomnia. Renal and urinary disorders: Frequent urination, UTI. Respiratory, thoracic and mediastinal disorders: Rhinitis, sinusitis, upper respiratory tract infection, cough, bronchitis. Skin and subcutaneous tissue disorders: Rash. Vascular disorders: Hot flushes. |
|
Patient Counseling Information
This drug may cause visual disturbances (e.g. blurred vision, decreased visual acuity, impaired depth perception), if affected, do not drive or operate machinery.
|
|
Drug Interactions
May increase risk of conduction disturbances and bronchoconstriction with concomitant use of β-adrenergic agonists. Additive effects with parasympathomimetic agents. Decreased metabolism and increased risk of adverse effects with inhibitors of CYP2D6 and CYP3A3/4.
|
|
Food Interaction
Decreased rate and extent of absorption with food.
|
|
Action
Description:
Mechanism of Action: Cevimeline is a cholinergic agonist that selectively binds to muscarinic receptors resulting in increased secretion of exocrine glands (e.g. salivary, sweat glands) and increased smooth muscle tone in gastrointestinal and urinary tracts. Pharmacokinetics: Absorption: Rapidly absorbed from the gastrointestinal tract. Decreased rate and extent of absorption with food. Time to peak plasma concentration: 1.5-2 hours. Distribution: Volume of distribution: Approx 6 L/kg. Plasma protein binding: <20%. Metabolism: Metabolised in the liver by CYP2D6 and CYP3A3/4 to cis and trans-sulfoxide, glucuronic acid conjugate, and N-oxide metabolites. Excretion: Mainly via urine (84% in 24 hours; 97% in 7 days); faeces (approx 0.5% in 7 days). Elimination half-life: 5±1 hour. |
|
Chemical Structure
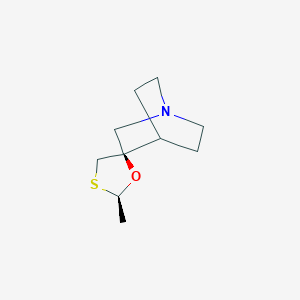 Source: National Center for Biotechnology Information. PubChem Database. Evoxac, CID=83898, https://pubchem.ncbi.nlm.nih.gov/compound/Evoxac (accessed on Mar. 25, 2020) |
|
Storage
Store between 20-25°C.
|
|
MIMS Class
|
|
ATC Classification
N07AX03 - cevimeline ; Belongs to the class of other parasympathomimetics.
|
|
References
Weber J, Keating GM. Cevimeline. Adis Data Information BV. 2008;68(12):1691-1698. Accessed 15/11/2019 Annotation of FDA Label for Cevimeline and CYP2D6. Pharmacogenomics Knowledgebase (PharmGKB). https://www.pharmgkb.org/. Accessed 15/11/2019. Anon. Cevimeline. Lexicomp Online. Hudson, Ohio. Wolters Kluwer Clinical Drug Information, Inc. https://online.lexi.com. Accessed 15/11/2019. Anon. CYP2D6-Cevimeline (Pharmacogenomics). Lexicomp Online. Hudson, Ohio. Wolters Kluwer Clinical Drug Information, Inc. https://online.lexi.com. Accessed 15/11/2019. Buckingham R (ed). Cevimeline Hydrochloride. Martindale: The Complete Drug Reference [online]. London. Pharmaceutical Press. https://www.medicinescomplete.com. Accessed 15/11/2019. Cevimeline Capsule (Lupin Pharmaceuticals, Inc.). DailyMed. Source: U.S. National Library of Medicine. https://dailymed.nlm.nih.gov/dailymed/. Accessed 15/11/2019.
|